Notation
In the case of an isentropic ideal gas, is the ratio of specific heats, known as the adiabatic index or as adiabatic exponent.
An isothermal ideal gas is also a polytropic gas. Here, the polytropic index is equal to one, and differs from the adiabatic index .
In order to discriminate between the two gammas, the polytropic gamma is sometimes capitalized, .
To confuse matters further, some authors refer to as the polytropic index, rather than . Note that
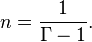
Read more about this topic: Polytropic Process