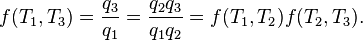Definition of Thermodynamic Temperature
The efficiency of the engine is the work divided by the heat introduced to the system or
- ,
where wcy is the work done per cycle. Thus, the efficiency depends only on qC/qH.
Because all reversible engines operating between the same heat reservoirs are equally efficient, any reversible heat engine operating between temperatures T1 and T2 must have the same efficiency, meaning, the efficiency is the function of the temperatures only:
In addition, a reversible heat engine operating between temperatures T1 and T3 must have the same efficiency as one consisting of two cycles, one between T1 and another (intermediate) temperature T2, and the second between T2andT3. This can only be the case if
Specializing to the case that is a fixed reference temperature: the temperature of the triple point of water. Then for anyT2and T3,
Therefore, if thermodynamic temperature is defined by
then the function f, viewed as a function of thermodynamic temperature, is
and the reference temperature T1 has the value 273.16. (Of course any reference temperature and any positive numerical value could be used—the choice here corresponds to the Kelvin scale.)
It follows immediately that
Substituting Equation 3 back into Equation 1 gives a relationship for the efficiency in terms of temperature:
Read more about this topic: Carnot's Theorem (thermodynamics)
Famous quotes containing the words definition of, definition and/or temperature:
“Scientific method is the way to truth, but it affords, even in
principle, no unique definition of truth. Any so-called pragmatic
definition of truth is doomed to failure equally.”
—Willard Van Orman Quine (b. 1908)
“No man, not even a doctor, ever gives any other definition of what a nurse should be than this—”devoted and obedient.” This definition would do just as well for a porter. It might even do for a horse. It would not do for a policeman.”
—Florence Nightingale (1820–1910)
“The bourgeois treasures nothing more highly than the self.... And so at the cost of intensity he achieves his own preservation and security. His harvest is a quiet mind which he prefers to being possessed by God, as he prefers comfort to pleasure, convenience to liberty, and a pleasant temperature to that deathly inner consuming fire.”
—Hermann Hesse (1877–1962)