Constant Pressure Heat Rejection (3-1)
The final stage (3-1) involves a constant pressure heat rejection back to the original state. From the first law of thermodynamics we find: .
From the definition of work:, we recover the following for the heat rejected during this process: .
As a result, we can determine the heat rejected as follows: 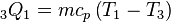 from the definition of constant pressure specific heats for an ideal gas:
from the definition of constant pressure specific heats for an ideal gas: 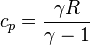 .
.
The overall efficiency of the cycle is determined by the total work over the heat input, which for a Lenoir cycle equals 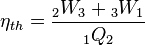 . Note that we gain work during the expansion process but lose some during the heat rejection process.
. Note that we gain work during the expansion process but lose some during the heat rejection process.
Read more about this topic: Lenoir Cycle
Famous quotes containing the words constant, pressure, heat and/or rejection:
“To keep up even a worthwhile tradition means vitiating the idea behind it which must necessarily be in a constant state of evolution: it is mad to try to express new feelings in a “mummified” form.”
—Alfred Jarry (1873–1907)
“Amid the pressure of great events, a general principle gives no help.”
—Georg Wilhelm Friedrich Hegel (1770–1831)
“Even if you find yourself in a heated exchange with your toddler, it is better for your child to feel the heat rather than for him to feel you withdraw emotionally.... Active and emotional involvement between parent and child helps the child make the limits a part of himself.”
—Stanley I. Greenspan (20th century)
“As between these two, the need that in its haste to be abolished cannot pause to be stated and the need that is the absolute predicament of particular human identity, one does not presume to suggest a relation of worth. Yet the distinction is perhaps not idle, for it is from the failure to make it that proceeds the common rejection as “obscure” of most that is significant in modern music, painting and literature.”
—Samuel Beckett (1906–1989)